 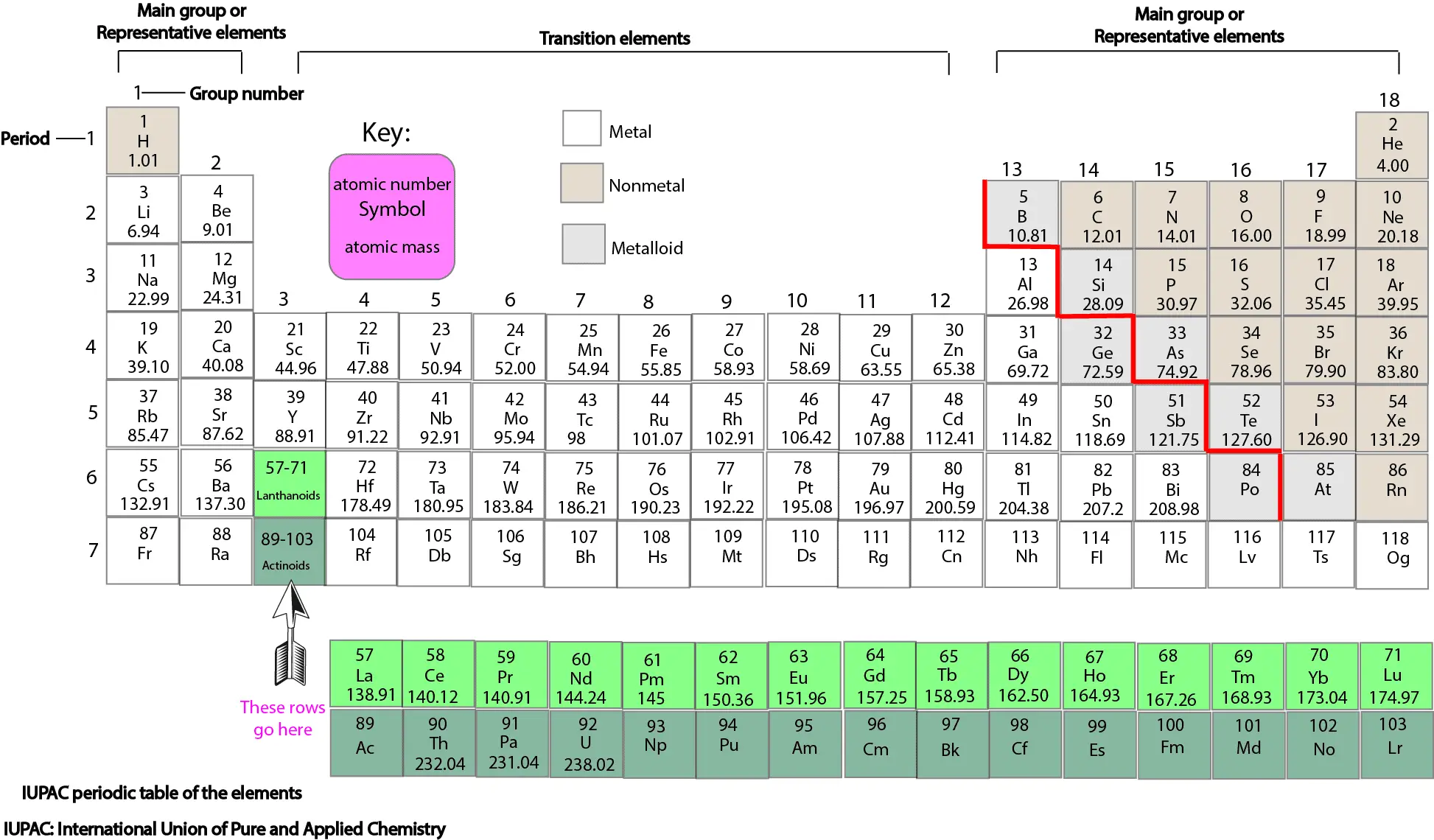
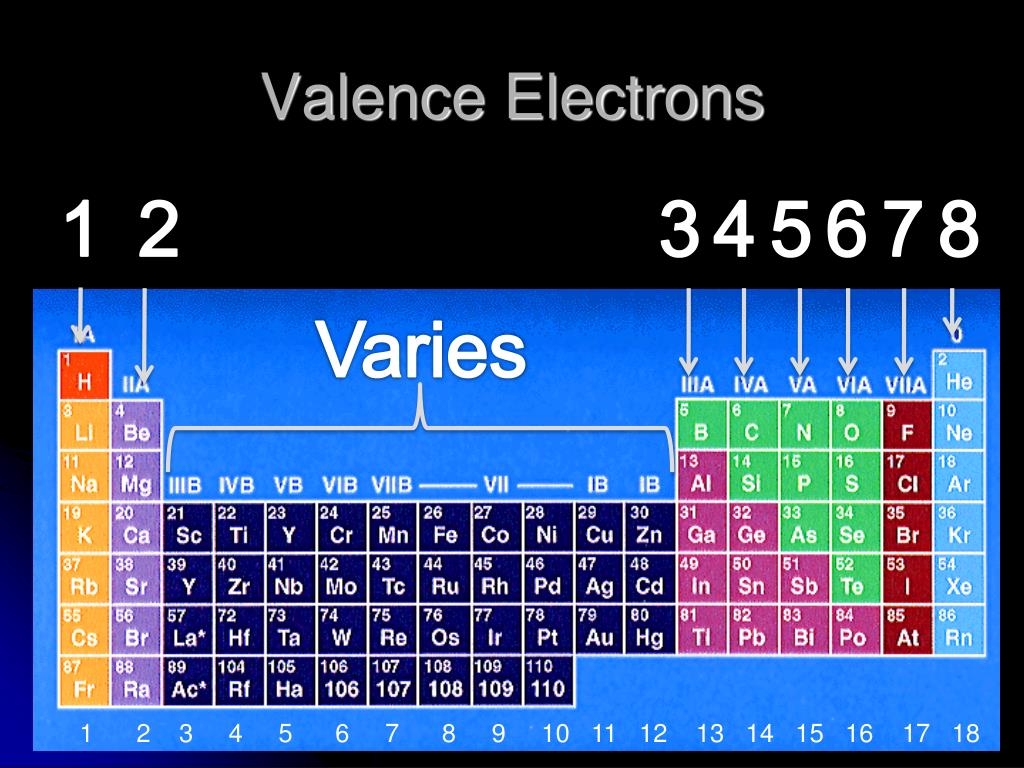
For example, iron can show two valencies, $2$ and $3$ in the compunds iron(II) sulphate ($\ce$, notice the difference that there is no plus or minus sign in valency). This works if you are using the definition of valence shell to be the outermost shell.īut it is wrong to apply the concept to determine the valency of the element, because that varies with the compound it is present in. Hence, magnesium has two valence electrons.The simplest answer is: d-block elements have number of valence electrons equal to their group number, which is equal to the number of electrons in the "valence shell". Now, the third subshell is the outermost shell with two electrons. On drawing the electrons for magnesium, you can see in the image below it has two electrons in the first subshell, eight electrons in the second, subshell and the remaining two will be in the third subshell. It has three shells and 12 electrons, which equals the atomic number of an element. The periodic table is arranged by atomic weight and valence electrons. Counting and addingĮlectrons from s, p, and d will give a total of 18 electrons in shell 3.Ĭonsider Magnesium.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
:max_bytes(150000):strip_icc()/PeriodicTableValence-58b5d8f95f9b586046df59fb.jpg)

 RSS Feed
RSS Feed
